Results
Filtros

Regulation of cultivation comes up for discussion at Anvisa after request for extension
10/06/2025The subject is on the agenda of the 6th Public Meeting of the Collegiate Board of Directors (Dicol) of 2025, scheduled for Wednesday (10/08)

Anvisa warns of deadline for comments on agenda including review of RDC 327/2019
Submission of registrations, videos, and requests for confidentiality ends at 11:59 pm this Friday (5); item revising rules for cannabis products will be defined at the Dicol meeting on December 10

Anvisa Collegiate Board has new composition defined after director's departure
Marcelo Mario Matos Moreira assumes as substitute in the Fourth Board, filling the vacancy of the former rapporteur of the review of RDC 327

Proposal to regulate cannabis cultivation is removed from Anvisa meeting agenda
Decision postpones vote on new rules for cultivating varieties with low THC content; new date for analysis has not been defined yet

Anvisa may vote on rules for national cultivation and control of cannabis this Wednesday (13)
Change may authorize cultivation by companies under strict control, with a limit of 0.3% THC; measure complies with a decision from the Superior Court of Justice (STJ) related to cultivation regulation.

Anvisa proposes creation of “Regulatory Sandbox” for cannabis cultivation by associations
Proposal aims to regulate the performance of non-profit entities in a controlled experimental environment, using the Regulatory Sandbox model to comply with STJ decision

Follow Live: Anvisa now debates revision of RDC 327 and cultivation
Anvisa directors meet this Wednesday (28) to discuss the update of medicinal cannabis regulations; learn how to follow

Anvisa discusses review of regulation of medicinal cannabis products
Public consultation proposal for the review of RDC No. 327/2019 will be evaluated by the Collegiate Board on December 19, 2024

Follow Live: Anvisa now debates review of RDC 327 and cultivation
Anvisa directors meet this Wednesday (28) to discuss the update of medicinal cannabis regulations; learn how to follow

Anvisa approves new Cannabis regulation and allows compounding pharmacies
Revision of RDC 327 allows the compounding sector to produce derivatives, while individual importation by individuals remains under old rules and will be analyzed separately

Proposal to restrict Cannabis importation (RDC 660) sparks reaction in the market
Proposal in the review of RDC 327/2019 to RDC 660/2022 divides opinions and raises doubts about access and judicialization

Voting on RDC 327 is postponed at Anvisa; ABIQUIFI evaluates new deadline as "legitimate instrument"
The Brazilian Association of Pharmaceutical Inputs Industry trusts that the request for a view by director Thiago Campos will result in a more in-depth analysis, maintaining the expectation of concluding the regulatory framework in January 2026

Anvisa suspends review of RDC 327 after director's request for further analysis
Rapporteur Rômison Mota advocated for changes impacting imports via RDC 660; new rule prioritizes national products when there is equivalence

Anvisa may vote on maintaining or removing the agenda item for the review of RDC 327/2019, which regulates medicinal cannabis products
The item revising RDC 327/2019 reaches a decisive week at Anvisa, and the sector awaits to know if the topic will proceed to a vote or be postponed once again
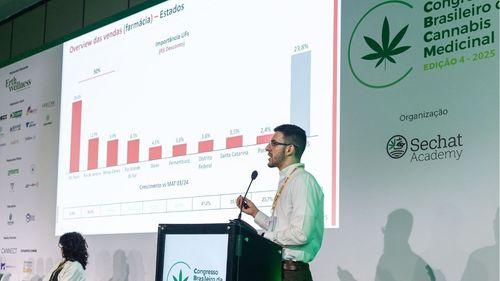
Cannabidiol grows over 70% in the pharmacy channel; Anvisa's RDC 327 review generates optimism in the sector
According to Filipe Campos, from Close-Up International, the market could expand even further with rule updates and greater medical adherence

“Extended deadline creates the possibility of presenting a more consistent proposal,” says Anvisa director about regulation of cultivation
Thiago Campos advocates for the extension of the regulation deadline and reinforces the importance of social listening in the process

Public participation in Anvisa's regulatory agenda continues until September 15
Public consultation allows society to express opinions on the agency's priorities for the next two years; preliminary list does not include cannabis

Anvisa removes the revision of RDC 327/2019 from the Collegiate Board meeting agenda
Update of the RDC included in the Regulatory Agenda 2024-2025 is expected for next year

RDC 327 review rapporteur bids farewell to Anvisa this Friday (19).
Rômison Rodrigues Mota leaves the position after a request for review delays the norm's vote; the topic will return to the regulatory agenda for 2026-2027

CFF advocates for magistral manipulation amid changes in Cannabis regulation
Entity reinforces that personalized preparations are essential when industrialized products do not meet clinical needs