Results
Filtros

Anvisa approves two cannabis-based products for pharmacies
The pharmaceutical company Farmausa Life Science has obtained registration for its Cannabidiol in two concentrations, 20 mg/mL and 100 mg/mL, with low THC content

Regulation of cannabis cultivation is the focus of Abiquifi's mission to Colombia, which started today in Bogotá
Brazilian delegation led by Abiquifi participates in a mission to Colombia to exchange experiences on the regulation of cannabis cultivation, with representatives from MAPA, Anvisa, Embrapa, and major pharmaceutical industries

Anvisa orders removal of cannabis products from CBD Brasil from the market
Anvisa has prohibited the manufacturing, sale, and promotion of CBD Brasil's cannabis products due to lack of sanitary authorization
STJ Minister sets final deadline for regulation of cannabis cultivation: March 2026
Minister Regina Helena Costa states that this will be the last extension and orders Anvisa to report compliance with each intermediate step of the plan

FNE promises to expedite cannabis exports to Brazil; agency seeks to understand Anvisa rules
Director-General of the National Narcotics Fund of Colombia, Milver Rojas, states that the process will be immediate once Anvisa confirms and foresees a return to the four to five-day deadline for each authorization

Univali is authorized by Anvisa to perform analyses of Cannabis products
New authorization expands the university's scope in Reblas and reinforces the quality control of plant derivatives in the national market
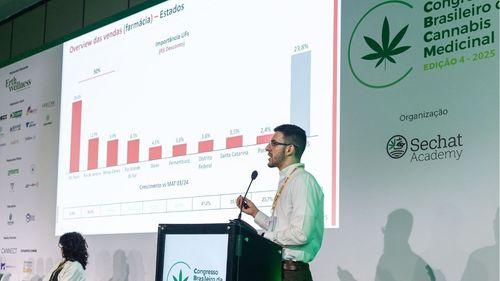
Cannabidiol grows over 70% in the pharmacy channel; Anvisa's RDC 327 review generates optimism in the sector
According to Filipe Campos, from Close-Up International, the market could expand even further with rule updates and greater medical adherence

IPT invests in new laboratory and aims at flower and extract analysis
Expansion of the institute in the sector, however, depends on new authorizations from Anvisa for the quality control of plant-based raw materials

DNA requests STJ authorization for regulatory sandbox after delay in hemp regulation
Company demands urgent measures in face of new missed deadlines by the government and Anvisa and proposes experimental environment under judicial supervision

Anvisa prohibits eight cannabis products due to lack of registration
The agency determined the suspension of the commercialization and advertising of all items from the brands Golden CBD Plus and Golden CBD+ Nanotech in the national territory.

Anvisa approves new cannabis-based product in Brazil
Pharmaceutical company Ephar has obtained registration for its Cannabidiol in two concentrations, 50 mg/mL and 100 mg/mL, which will be marketed in a low THC content drop solution.

Anvisa prohibits cannabis products from two companies for irregular sale; responsible parties claim to operate within the rules
Regulatory agency bans commercialization and advertising of items from the brands due to lack of sanitary authorization; understand the decision and the companies' positions

Anvisa prohibits cannabis products from Santa Catarina company due to lack of authorization
Regulatory agency determines the seizure of all batches from the company and another distributor due to irregularities in registration and manufacturing

The path of cannabis in animal well-being is made of science, empathy, and new hopes
The use of medicinal cannabis in pets has been growing in Brazil since Anvisa's authorization in 2024, bringing relief for chronic pain, epilepsy, and anxiety in animals, and renewed hope for pet owners

“Extended deadline creates the possibility of presenting a more consistent proposal,” says Anvisa director about regulation of cultivation
Thiago Campos advocates for the extension of the regulation deadline and reinforces the importance of social listening in the process

Anvisa approves new cannabis-based product in Brazil
Cannten's extract with 71.33 mg/mL expands medicinal treatment options in the country and receives authorization for commercialization

Regulation of cultivation comes up for discussion at Anvisa after request for extension
10/06/2025The subject is on the agenda of the 6th Public Meeting of the Collegiate Board of Directors (Dicol) of 2025, scheduled for Wednesday (10/08)

Anvisa approves new pharmaceutical product based on cannabis in Brazil
Extract from the company Endogen expands the portfolio of options for medicinal treatments and already has sanitary authorization for commercialization

One year after Anvisa's approval, MAPA still does not regulate veterinary cannabis
Without defined rules, veterinarians face legal uncertainty and animals still have limited access to medicinal cannabis treatment

Government asks STJ for additional 180 days to deliver regulation on cannabis cultivation
Ministry of Health and Anvisa claim technical complexity and propose a new schedule that extends until March 2026 to define the rules