Results
Filtros

Company obtains decision from Federal Court and is authorized to handle cannabis products
Favorable decision to Biomagistral network allows the production of formulations with Cannabidiol under prescription

Study indicates that cannabis use has surpassed cigarette use among Americans
The data reveals a cultural shift that reflects new perceptions about health, habits, and public policies

Meta deletes 47 cannabis-related profiles and sparks debate about platform policies
According to lawyer Clayton Medeiros, complaints and changes in the Civil Framework may have driven the action; political pressure ensured the return of some profiles

Government asks STJ for additional 180 days to deliver regulation on cannabis cultivation
Ministry of Health and Anvisa claim technical complexity and propose a new schedule that extends until March 2026 to define the rules
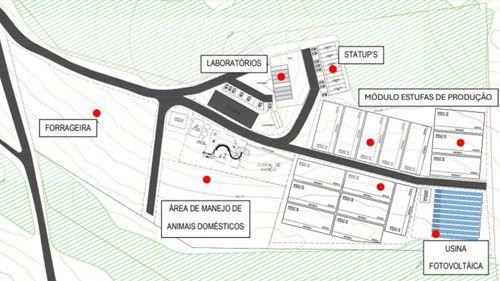
UNESP inaugurates nucleus for research and cultivation of cannabis in a 5.5-hectare area
NuDeCann will have modular expansion in three phases until 2028, focusing on science, health, and sustainability

100% of patients with insomnia slept longer with cannabis in study by Fiocruz and APEPI
Oil rich in CBD and THC also reduced the time to fall asleep in 87.5% of participants, who no longer responded to conventional treatments

More than 500 companies offer cannabis products to Brazil through importation; products now exceed 600
Anvisa points out the USA as the main supplier, followed by Canada, Colombia, and Uruguay

Logistics, Regulation, and Security: The Impasse on Storing Cannabis in Brazil
In Deusa Cast, experts discuss the possibility of storing cannabis-based products in Brazil. Regulatory advances, legal obstacles, and logistical impacts were debated in light of RDCs 660 and 327

Logistics, Regulation, and Security: The Impasse on Storing Cannabis in Brazil
In Deusa Cast, experts discuss the possibility of storing cannabis-based products in Brazil. Regulatory advances, legal hurdles, and logistical impacts were debated in light of RDCs 660 and 327

Reblas Network includes cannabis product analysis; Brazil has 16 accredited laboratories
Published by Anvisa, new resolution expands the scope of the network of analytical laboratories and reinforces the quality control of cannabis-based products in the country

Reblas Network includes cannabis product analysis; Brazil has 16 accredited laboratories
Published by Anvisa, the new resolution expands the scope of the network of analytical laboratories and reinforces the quality control of cannabis-based products in the country

Reblas Network includes cannabis product analysis; Brazil has 16 accredited laboratories
Published by Anvisa, the new resolution expands the scope of the network of analytical laboratories and reinforces the quality control of cannabis-based products in the country

Netherlands and Cannabis Seed Banks: Pioneering that Shaped the Global Market
From Amsterdam's coffeeshops to Brazilian genetic banks, the history of cannabis shows how the Netherlands became a global reference in innovation

Use of Cannabis sativa advances in small animal veterinary medicine, Brazilian study finds
Brazilian research highlights the benefits of Cannabis sativa in the treatment of dogs and cats, with applications in chronic diseases, neurological conditions, and pain management

Associations may have more space in the debate on the regulation of cannabis cultivation in Brazil
Entrepreneur Fabrício Penafiel considers the inclusion of practical experience of producers in the proposal, with a deadline set for September 30th, a historic opportunity.

Request for cannabis-based medicine without registration at Anvisa must be judged by the Federal Justice
STJ decision reinforces federal competence for actions involving unregistered drugs at the regulatory agency

Request for cannabis-based medicine without registration at Anvisa must be judged by the Federal Court
STJ decision reinforces federal jurisdiction for actions involving unregistered drugs at the regulatory agency

Experience with Daughter Inspires Brazilian Scientist to Lead Innovative Study on Cannabis for Autism
Revivid invested in the groundbreaking research of scientist and mother Micheline Donato, who turned her experience with her daughter's autism into a clinical study on the use of cannabis in adults.

Anvisa concludes public consultation on new regulation of Cannabis products
Deadline for submitting contributions to the proposal for the revision of RDC 327/2019 ended this Monday (2). Participation exceeded 7,000 forms

Ministry of Health works to regulate cannabis cultivation and distribution through the SUS
General coordinator details the efforts made by the public agency