Reblas Network includes cannabis product analysis; Brazil has 16 accredited laboratories
Published by Anvisa, the new resolution expands the scope of the network of analytical laboratories and reinforces the quality control of cannabis-based products in the country
Published on 09/11/2025

The measure was confirmed by Resolution No. 3,459, published on September 8 in the Official Gazette. Image: Canva pro
Analytical laboratories, whether public or private, accredited by the National Health Surveillance Agency (Anvisa), are officially authorized to perform analyses of cannabis products. The measure was confirmed by Resolution No. 3,459, published on September 8 in the Official Gazette.
The resolution modifies the scope of the Analytical Quality Center Ltd., a member of the Brazilian Network of Analytical Laboratories in Health (Reblas), officially including the category of cannabis products.
As a result, the laboratory now offers quality control services for products derived from the plant, as per Art. 5 of Resolution RDC No. 928/2024:
"Art. 5 Laboratories providing services that perform quality control tests on batches of finished products must be accredited in Reblas in scopes corresponding to the respective categories of products analyzed."
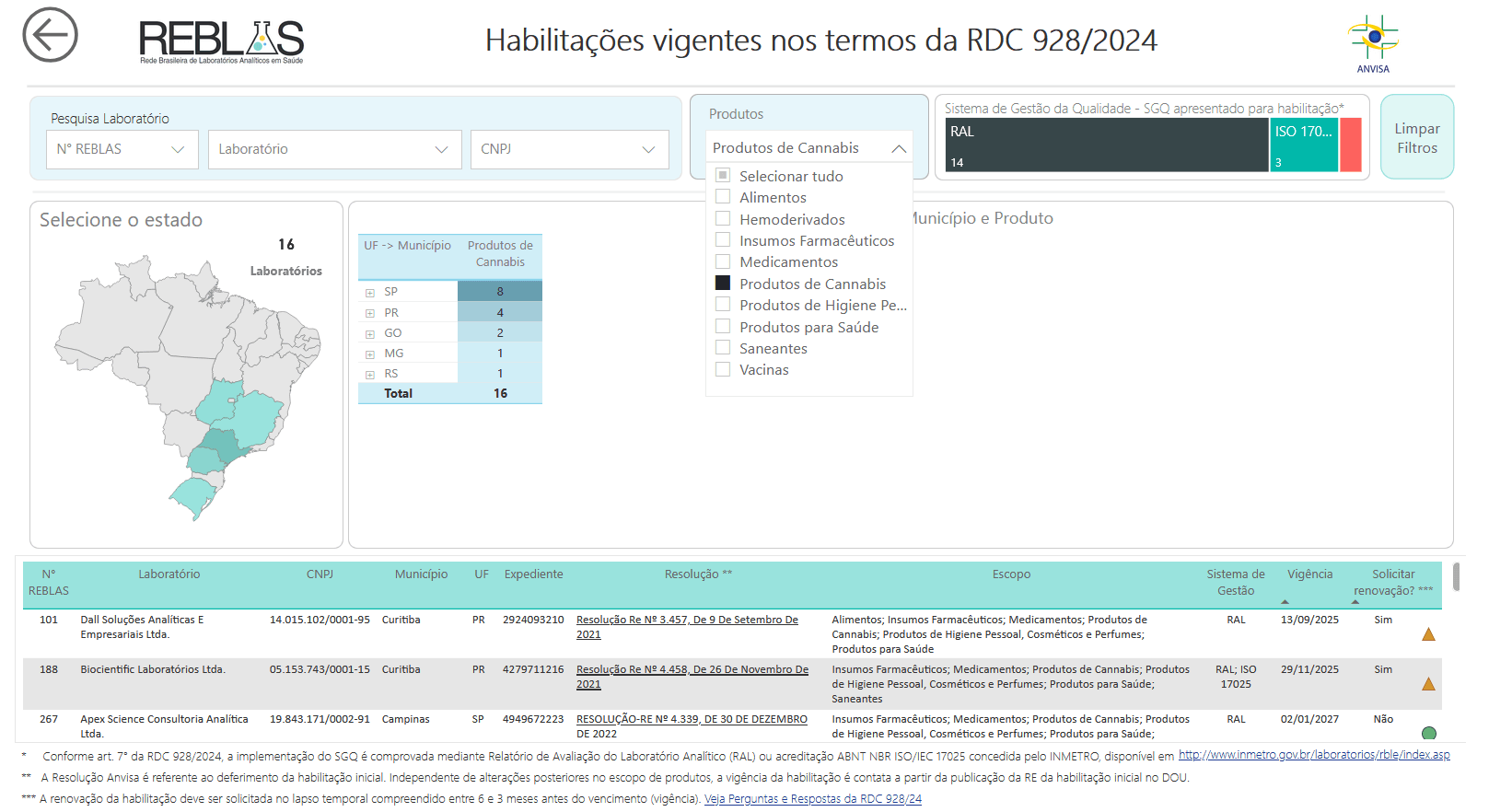
16 laboratories are already accredited
Reblas brings together analytical laboratories, public and private, accredited by Anvisa to offer sanitary services of interest with quality, safety, reliability, and traceability.
Currently, 16 laboratories already have valid accreditation for cannabis product analysis. The state of São Paulo concentrates half of them, followed by Paraná (4), Goiás (2), Minas Gerais (1), and Rio Grande do Sul (1). All accreditations were granted by Anvisa resolutions.
The scientific director of Sechat, Dr. Pedro Pierro, highlighted the importance of the measure. “The ability to ensure the quality of cannabis-based products through RDC 660, with the support of Reblas laboratories, is essential for doctors and patients.”
An example cited is Carmen's Medicinals, which in 2024 obtained validation attesting that their oils have a high degree of purity and quality. According to the company, this provides security to doctors and patients that they are prescribing and consuming products that meet the most rigorous standards established by Anvisa.
Ricardo Pettená, the company's executive director, emphasizes that the investment in quality is directly reflected in the final product. “Having the Reblas certificate conveys credibility and security to the medical community when prescribing,” he states.
For him, certification also helps differentiate the “wheat from the chaff,” encouraging the market to adapt and offer products from RDC 660 with standards equal to or even higher than those of RDC 327. “Previously, we only worked with the COA certificate, which, we understand, does not have the same credibility with the market and Anvisa itself. Reblas highlights which companies truly have a commitment to quality and which do not,” he concludes.









